Interactive niches for hematopoietic stem cells based on cell-secreted and synthetic polymer matrices
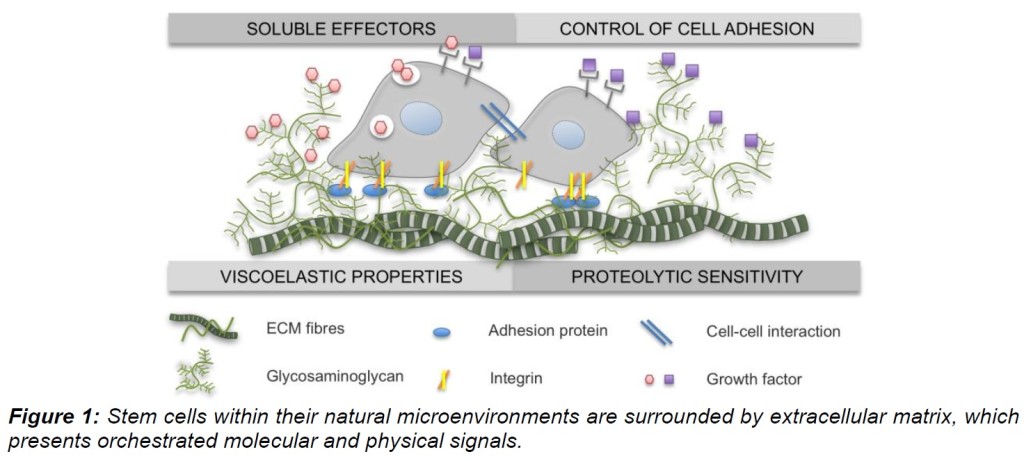
The principal components of the hematopoietic stem cell (HSC) niche have been intensively studied in the last decade. As reviewed recently, cellular components as well as matrix-bound morphogens have been shown to be involved in the regulation of HSC homeostasis, mobilisation and homing after transplantation (Ehninger et al., 2011).
The project aims at developing three-dimensional (3D) polymer culture systems to exogenously control expansion and differentiation of human hematopoietic stem and progenitor cells (HSPC) ex vivo. Emphasis is put on the interaction between selected extracellular matrix (ECM) components and HSPC. Recently, we established a platform of surface-immobilized ECM preparations derived from decellularized MSC and demonstrated their supportive role for the expansion of MSC and HSPC as confirmed in long-term engraftment in vivo experiments.
Accordingly, we plan to (i) perform targeted RNA-interference within primary MSC to be grown in culture for matrix production in order to identify candidate proteins that determine adhesion, migration and expansion of HSPC. Furthermore, MSC culture conditions and matrix processing schemes will be extended to modulate the characteristics of the obtained matrices. In parallel, we will (ii) customize a novel set of in situ assembling poly(ethylene glycol)-heparin hydrogels by adjusting elasticity, enzymatic (cell-driven) degradability and presentation/release of functional (ECM-derived) peptides and growth factors including SDF-1, SCF, MCAM and Angiopoietin-like proteins. HSPC grown in culture with genetically targeted ECM preparations or embedded in a library of biohybrid polymer gels will be thoroughly characterized in vitro. Cells expanded under the most effective conditions will be tested for their long-term engraftment potential using a previously established xenograft model.
Participating group members:
Martin Bornhaeuser, Manja Wobus, Martin Kraeter, Katrin Mueller, Kristin Heidel